 Scaling Up
Mr Sarwar, who had the idea for the business five years ago when he visited Babson College near Boston with Adam Hunter, now Phlo Chief Commercial Officer, said the plan is to scale up “city by city”.
He commented:
“There are 36 million patients on prescription in England. About 1% have gone online. My firm belief is the next five million patients will go online in the next two years – it is our job to go and grab a nice slice of that market share.”
From launch last year, demand and patient numbers have been growing consistently, but in the period since COVID-19 became a significant public health issue, growth has exceeded all expectations.
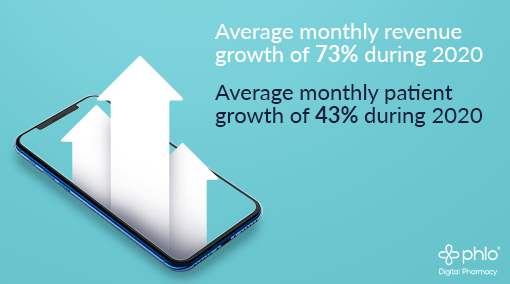
Since the start of the year, Phlo has seen an impressive average 73% growth in MOM revenue, 43% average growth in MOM patient numbers and a 93% uplift in staff numbers. Phlo has serviced over 5000 patients to date from Jersey to Orkney.
Phlo is headquartered in Glasgow and currently employs 32 members of staff across high-value pharmacy and technology roles. The Pharmacy operations team is situated in central London.
First Digital Pharmacy Crowdfund in UK
This is the first UK Digital pharmacy crowdfund and interest has been high. The initial target of £1.65m was hit in the first 24 hours and the campaign is set to continue until 11thNovember 2020.
Mr Sarwar commented: “I’ve been blown away by the level of support we have received both from our patients and the public. The funds raised from this crowdfunding round will help us to expand our operations in major cities in England and allow us to continue on our path to building the most technology advanced digital pharmacy platform in the UK”
Phlo has also received investment from the UK Government Future Fund in the form of a convertible loan note. Investments will pay 8% interest until they are converted to shares at the completion of a follow – up investment round. Longer term, the company is eyeing up venture capital funding.
To find out more about Phlo’s crowdfunding opportunity, please visit www.crowdcube.com/phlo.
Scaling Up
Mr Sarwar, who had the idea for the business five years ago when he visited Babson College near Boston with Adam Hunter, now Phlo Chief Commercial Officer, said the plan is to scale up “city by city”.
He commented:
“There are 36 million patients on prescription in England. About 1% have gone online. My firm belief is the next five million patients will go online in the next two years – it is our job to go and grab a nice slice of that market share.”
From launch last year, demand and patient numbers have been growing consistently, but in the period since COVID-19 became a significant public health issue, growth has exceeded all expectations.
Since the start of the year, Phlo has seen an impressive average 73% growth in MOM revenue, 43% average growth in MOM patient numbers and a 93% uplift in staff numbers. Phlo has serviced over 5000 patients to date from Jersey to Orkney.
Phlo is headquartered in Glasgow and currently employs 32 members of staff across high-value pharmacy and technology roles. The Pharmacy operations team is situated in central London.
First Digital Pharmacy Crowdfund in UK
This is the first UK Digital pharmacy crowdfund and interest has been high. The initial target of £1.65m was hit in the first 24 hours and the campaign is set to continue until 11thNovember 2020.
Mr Sarwar commented: “I’ve been blown away by the level of support we have received both from our patients and the public. The funds raised from this crowdfunding round will help us to expand our operations in major cities in England and allow us to continue on our path to building the most technology advanced digital pharmacy platform in the UK”
Phlo has also received investment from the UK Government Future Fund in the form of a convertible loan note. Investments will pay 8% interest until they are converted to shares at the completion of a follow – up investment round. Longer term, the company is eyeing up venture capital funding.
To find out more about Phlo’s crowdfunding opportunity, please visit www.crowdcube.com/phlo. UK on-demand digital pharmacy Phlo surpasses £1.65m funding target on Crowdcube
 Scaling Up
Mr Sarwar, who had the idea for the business five years ago when he visited Babson College near Boston with Adam Hunter, now Phlo Chief Commercial Officer, said the plan is to scale up “city by city”.
He commented:
“There are 36 million patients on prescription in England. About 1% have gone online. My firm belief is the next five million patients will go online in the next two years – it is our job to go and grab a nice slice of that market share.”
From launch last year, demand and patient numbers have been growing consistently, but in the period since COVID-19 became a significant public health issue, growth has exceeded all expectations.
Since the start of the year, Phlo has seen an impressive average 73% growth in MOM revenue, 43% average growth in MOM patient numbers and a 93% uplift in staff numbers. Phlo has serviced over 5000 patients to date from Jersey to Orkney.
Phlo is headquartered in Glasgow and currently employs 32 members of staff across high-value pharmacy and technology roles. The Pharmacy operations team is situated in central London.
First Digital Pharmacy Crowdfund in UK
This is the first UK Digital pharmacy crowdfund and interest has been high. The initial target of £1.65m was hit in the first 24 hours and the campaign is set to continue until 11thNovember 2020.
Mr Sarwar commented: “I’ve been blown away by the level of support we have received both from our patients and the public. The funds raised from this crowdfunding round will help us to expand our operations in major cities in England and allow us to continue on our path to building the most technology advanced digital pharmacy platform in the UK”
Phlo has also received investment from the UK Government Future Fund in the form of a convertible loan note. Investments will pay 8% interest until they are converted to shares at the completion of a follow – up investment round. Longer term, the company is eyeing up venture capital funding.
To find out more about Phlo’s crowdfunding opportunity, please visit www.crowdcube.com/phlo.
Scaling Up
Mr Sarwar, who had the idea for the business five years ago when he visited Babson College near Boston with Adam Hunter, now Phlo Chief Commercial Officer, said the plan is to scale up “city by city”.
He commented:
“There are 36 million patients on prescription in England. About 1% have gone online. My firm belief is the next five million patients will go online in the next two years – it is our job to go and grab a nice slice of that market share.”
From launch last year, demand and patient numbers have been growing consistently, but in the period since COVID-19 became a significant public health issue, growth has exceeded all expectations.
Since the start of the year, Phlo has seen an impressive average 73% growth in MOM revenue, 43% average growth in MOM patient numbers and a 93% uplift in staff numbers. Phlo has serviced over 5000 patients to date from Jersey to Orkney.
Phlo is headquartered in Glasgow and currently employs 32 members of staff across high-value pharmacy and technology roles. The Pharmacy operations team is situated in central London.
First Digital Pharmacy Crowdfund in UK
This is the first UK Digital pharmacy crowdfund and interest has been high. The initial target of £1.65m was hit in the first 24 hours and the campaign is set to continue until 11thNovember 2020.
Mr Sarwar commented: “I’ve been blown away by the level of support we have received both from our patients and the public. The funds raised from this crowdfunding round will help us to expand our operations in major cities in England and allow us to continue on our path to building the most technology advanced digital pharmacy platform in the UK”
Phlo has also received investment from the UK Government Future Fund in the form of a convertible loan note. Investments will pay 8% interest until they are converted to shares at the completion of a follow – up investment round. Longer term, the company is eyeing up venture capital funding.
To find out more about Phlo’s crowdfunding opportunity, please visit www.crowdcube.com/phlo. WPP reveals drop in Q3 revenue
Lloyds returns to profit amid housing boom
BT raises profit guidance despite 20% fall in profits
Three reasons why New Year’s Eve may be an eventful time for equities
The Christmas COVID Crunch
Despite the police commissioner’s misguided tough guy comments about breaking up gatherings in breach of COVID rules, it seems almost inevitable that families across the Western world will come together to celebrate Christmas. And, with this in mind, we can make the assumption that the days following Christmas will be filled with headlines fretting about spikes in cases. Of course, the extent to which this comes to pass will be influenced by the lockdown measures in place over the festive period. However, many Twitter users in the UK and US have been vocal about their intentions to see loved ones, regardless of official restrictions. Should this be the case, we can expect further downside for Western equities. Politicians may be keen to avoid stringent lockdown measures over Christmas, but this might require tougher measures before – and, importantly – after the holiday period. While new lockdown measures between now and Christmas are already being priced in by shaky indexes this week, the severity of new case numbers will have a hand in steering the range of restrictions in place as we enter the New Year.Equities will love the duck no longer being lame
In a roundabout way: the new US president will stop being a lame duck in January. Save for the nightmare outcome of a close result in the presidential election next week, the next US president will be inaugurated in January. At present, bookies have three likely outcomes – a Democrat clean sweep, a Biden win but mixed legislature, and a Trump win with mixed legislature. Now, both candidates look likely to implement a new stimulus package once they take office, and whoever is in power will normally drop some breadcrumbs about what new support measures will look like ahead of time. Certainly, there have been reports saying that markets have priced in a Biden win next Tuesday, and such an outcome would likely result in more generous stimulus being introduced in the New Year. While markets would rub their hands eagerly at such a prospect, they’ll recoil in disgust at his new taxation policy plan. Trump will implement more modest stimulus, and thus the excitement for new support, while present, while be more muted. In contrast, Biden will likely bring in more extensive support, but – and despite BlackRock debunking some of the practical fears being raised – the giddy sentiment this incites will to some extent be cancelled out by equities pricing in heavier taxation.New Year, new hope
Perhaps the variable offering the strongest potential upside is the ultimate light at the end of the tunnel: a vaccine. With status updates being posted this week on the potential efficacy of the Oxford-AstraZeneca vaccine candidate, and Pfizer flexing the muscles of its vast vaccine roll-out logistics operations, any hope of there being an end in sight should have been rekindled. Indeed, just today, GSK and Sanofi announced a joint Statement of Intent to supply 200 million doses of its vaccine to the COVAX initiative, once approval has been received. Regarding a timeframe for these promising words to become reality, GSK hopes to achieve regulatory approval within the first half of 2021, and their effort doesn’t even seem to be the current front-runner. With that in mind, we may be on the brink of a vaccine candidate being made publicly available in the early stages of 2021 – or at least, that’s what’s currently being alluded to. Failing this, we’ll no doubt have more sweet nothings to sate our appetite for good news. And, if nothing else, some promising updates at the turn of the New Year may be enough to leave equities in a good mood.Hogma-nay or Hogma-yay?
I’m opinionated but not an oracle, and I won’t give you a clear answer I’ll regret at a later date. What I will say, though, is that you take time off over the holiday period at your peril. Even with just the three themes we’ve discussed, family arguments around the dinner table are unlikely to be the main sites of drama as the year draws to a close.UPS delivers 16% revenue growth but shares price in future downside
Ibstock reports decline in revenue, shares fall
Boeing to cut 20% total workforce
GSK and Sanofi sign Statement of Intent to provide 200m COVID vaccines
Once necessary approvals are gained, both companies say they will make their adjuvanted recombinant protein-based COVID-19 vaccine available to the COVAX Facility, to contribute towards its goal of “[reaching] those in need, whoever they are and wherever they live”.
Speaking on the companies’ commitment, Thomas Triomphe, Executive Vice President and Global Head of Sanofi Pasteur, said:
“To address a global health crisis of this magnitude, it takes unique partnerships. The commitment we are announcing today for the COVAX Facility can help us together stand a better chance of bringing the pandemic under control. This moment also reflects our long-term commitment to global health and ensures our COVID-19 vaccines are affordable and accessible to those most at risk, everywhere in the world.”
Roger Connor, President of GSK Vaccines added:
“Since we started working on the development of COVID-19 vaccines, GSK has pledged to make them available to people around the world. We are proud to be working with Sanofi to make this adjuvanted recombinant protein-based vaccine available to the countries signed up to the COVAX Facility as soon as possible – this has the potential to be a significant contribution to the global fight against COVID-19.”
Having initiated the first of two phases of its trials on September 3, with 440 participants enrolled, GSK stated that it expects first results in early December 2020. It said these results being secured would be ‘pivotal’ to support the initiation of a Phase 3 study before the end of the year. Should the data prove sufficient for licensure application, GSK said it will request regulatory approval during the first half of 2021. In parallel, GSK said that it and Sanofi had been scaling up manufacturing of the antigen and adjuvant respectively. The company adds that the use of an adjuvant technology is particularly important in a pandemic situation, given that it may reduce the amount of vaccine protein required per dose. It continued, saying that it does not expect profit to be made from the COVID vaccine during the pandemic phase, and said it will invest any short-term profit back into COVID-related research and long-term vaccine preparedness.