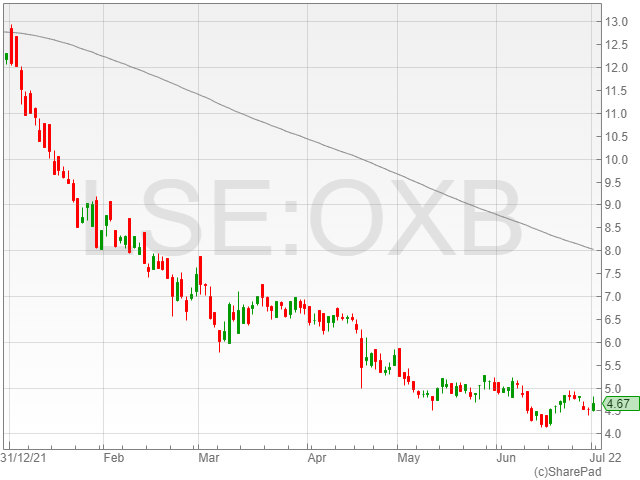
Oxford Biomedica shares were up 3% to 468p in late morning trading on Friday after the firm announced it had entered into a three-year Master Services and Development Agreement with AstraZeneca.
The Agreement would reportedly facilitate the potential future manufacturing opportunities for the AstraZeneca Covid-19 vaccine.
Oxford Biomedica noted the deal represents an expansion of the original Master Supply and Development Agreement announced between both groups in September 2020.
The working relationship is set to have Oxford Biomedica complete its manufacture of AstraZeneca Covid-19 vaccines in Q4 2022, followed by Oxford Biomedica providing the use of its 84,000 square foot manufacturing facility for AstraZeneca to take advantage of on an as needed basis after 2022.
Oxford Biomedica confirmed an expected recognised revenue of approximately £30 million from AstraZeneca in the current financial year in line with the terms of the original Agreement and inclusive of all batches of vaccine manufactured in HY1 2022.
“I am delighted that our close partnership with AstraZeneca has been extended. I am proud of the work of all our colleagues at Oxford Biomedica that has enabled us to deliver more than 100 million doses of lifesaving COVID-19 vaccine,” said Oxford Biomedica CEO Dr. Roch Doliveux.
“While contributing to the efforts to fight the pandemic, this has also demonstrated Oxford Biomedica’s ability to expand the scope of our innovative process development services and deliver high-performing manufacturing solutions beyond lentiviral vectors.”
“We look forward to continuing to work closely with AstraZeneca and execute on our strategy to become a global leader across all viral vectors, enabling Cell and Gene Therapy companies to deliver their life-changing therapies to patients.”
